SyncBox



Synchronizes the clock of BrainAmp MR amplifiers with the master clock driving the MRI scanner’s gradient system
Significantly reduces timing related errors and boosts the quality of gradient artifact correction
SyncBox scanner interface and scanner clock output are physically connected (using galvanic coupling) to avoid potential influence on the MR scanner system
Seamless integration with BrainVision Recorder and Analyzer software by means of dedicated trigger codes and implemented algorithms
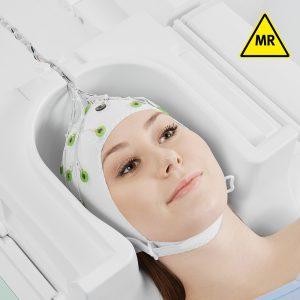
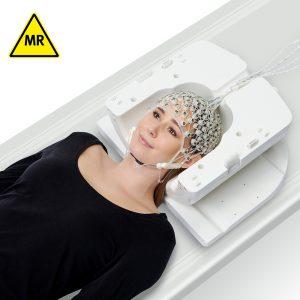
SyncBox: Must-have solution to boost data quality for concurrent EEG and fMRI recordings
Phase synchronization between the EEG equipment and the MRI scanner results in temporal stability of the EEG acquisition in relation to the switching of the gradients during the MR acquisition. This leads to significant improvement in the quality of gradient artifact correction and therefore the recorded data quality.
Reference: Mandelkow, H., Halder, P., Boesiger, P., & Brandeis, D. (2006). Synchronization facilitates removal of MRI artefacts from concurrent EEG recordings and increases usable bandwidth. Neuroimage, 32(3), 1120-1126. https://doi.org/10.1016/j.neuroimage.2006.04.231
The SyncBox is essential to recording high quality EEG data in the MR environment and is it compatible with all major commercial MRI scanners available on the market.
| SyncBox | Technical specifications |
|---|---|
| SyncBox Main Unit | |
| Input voltage | 5 V DC |
| Current consumption (via USB) | < 150 mA |
| Properties of the input signal | 0.4 Vpp to 5 Vpp, hysteresis approx. 150 mV |
| Frequency of the input signal | Max. 30 MHz |
| Input impedance (on the scanner side) | 50 Ω, BNC socket |
| Required input signal shape | Sine or square |
| Computer interface | USB 2.0 |
| Length of the connecting cable | Max. 20 m |
| Suitability for use in the scanner room | No, but can be used in the control room |
| Dimensions | Approx. 132 mm x 70 mm x 30 mm (without connections) |
| Weight | Approx. 160 g |
| SyncBox Scanner Interface | |
| Power supply | Via SyncBox Main Unit |
| Input (on the scanner side) | 50 Ω, BNC socket with 30cm BNC cable; available in both SyncBox Scanner Interface versions Optical Input for single optical fiber, connector AFBR-2624; available in SyncBox Scanner Interface Duo version only |
| Output | 50 Ω, BNC socket |
| Galvanic isolation | Yes, dielectric strength > 4 kV |
| Length of the connecting cable | 30 cm |
| Dimensions | Approx. 90 mm x 45 mm x 25 mm |
| Weight | Approx. 80 g |
| Miscellaneous | |
| Suitable for use in MR scanner room | No |
| Medical device | No |
| CE marking | The Brain Products GmbH confirms the electromagnetic compatibility (EMC) of this product according to the Directive 2014/30/ EU of the European Parliament and of the Council of 26 February 2014 on the harmonisation of the laws of the Member States relating to electromagnetic compatibility. The Brain Products GmbH confirms the RoHS compliance of this product according to Directive 2011/65/EU of the European Parliament and the Council of 8 June 2011 on the restriction of the use of certain hazardous substances in electrical and electronic equipment (recast published in the Official Journal of the European Union on 1 July 2011) as well as all its amendments up to and including the Commission delegated directive (EU) 2015/863 of 31 March 2015 (published in the 0fficial Journal of the European Union on 4 June 2015). |
Recommended add-ons