BrainVision CapTrak




Professional camera-based electrode location scanning system
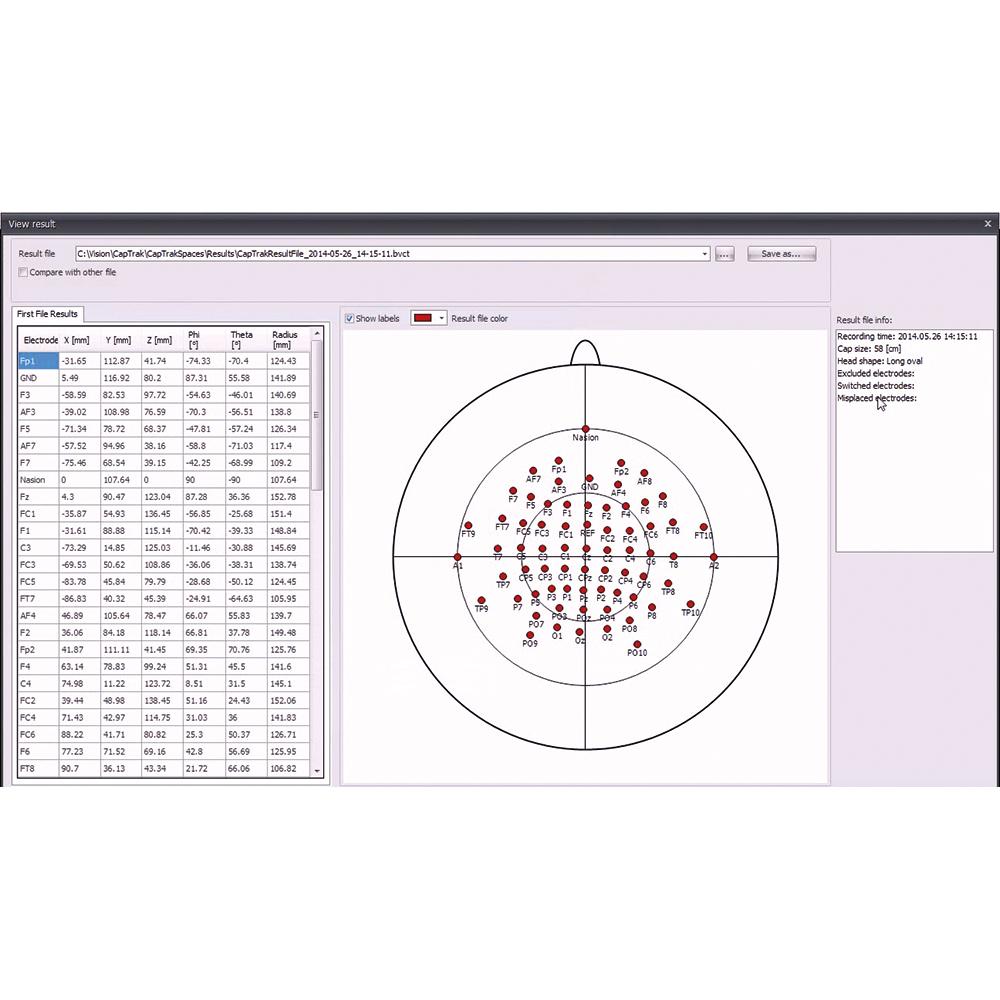
Accurate optical electrode detection
Audio-visual guided registration process
Intuitive software for fast electrode localization (128 electrodes in 5 minutes)
BrainVision CapTrak: Fast and accurate electrode localization system for active electrodes
 Fast: Scan 128 electrode positions in 5 minutes; works with actiCAP slim (and previous actiCAP) electrodes.
Fast: Scan 128 electrode positions in 5 minutes; works with actiCAP slim (and previous actiCAP) electrodes.- Accurate: Scan up to 160 individual electrodes with a mean deviation of less than 1 mm.
- Reliable: Use the BrainVision CapTrak software to compare measurements and determine if the cap was moved between scans.
- Smart: Let CapTrak alert you when it detects mismatches in the expected electrode locations.
- Flexible:
→ Compatible with actiCAP ControlBox (BrainAmp), actiCHamp (Plus) and LiveAmp.
→ Select the format of the coordinate file and further process the data in BrainVision Analyzer 2 or other software.
The system comes ready to use out of the box and includes a light, hand-held scanner equipped with two integrated cameras, intuitive software and a pre-configured computer.

By loading the video, you agree to YouTube's privacy policy.
Learn more
The BrainVision CapTrak technical specifications and software system requirements are listed in the table below.
You can review the Brain Products Software License Agreement here.
| CapTrak | Technical specifications |
|---|---|
| Scanner data | |
| Connection | USB 3.0 |
| Supply voltage and power | Max. 4 W (at 5 VDC) |
| Dimensions (L/H/D) | 28 cm x 18.5 cm x 13 cm |
| Weight | Approx. 840 g |
| Software System Requirements | |
| Operating system | Windows® 10 |
| RAM | 4 GB |
| Available hard-disc space | 70 MB |
| CPU | 3.4 GHz |
| USB | USB 3.0 or higher |
| Miscellaneous | |
| Suitable for use in MR scanner room | No |
| Medical device | No |
| CE marking | The Brain Products GmbH confirms the electromagnetic compatibility (EMC) of this product according to the Directive 2014/30/EU of the European Parliament and of the Council of 26 February 2014 on the harmonisation of the laws of the Member States relating to electromagnetic compatibility. The Brain Products GmbH confirms the RoHS compliance of this product according to Directive 2011/65/EU of the European Parliament and the Council of 8 June 2011 on the restriction of the use of certain hazardous substances in electrical and electronic equipment (recast published in the Official Journal of the European Union on 1 July 2011) as well as all its amendments up to and including the Commission delegated directive (EU) 2015/863 of 31 March 2015 (published in the Official Journal of the European Union on 4 June 2015). |